
Esme Anderson Grant
Project Title
Neuroprotection of Retinal Ganglion Cells by Photobiomodulation
Chief investigator
Professor Robert Casson
Professor Robert Casson MB,BS (Hons), M.Biostatistics, DPhil, FRANZCO heads the Ophthalmic Research Laboratory in the Discipline of Ophthalmology and Visual Science at Adelaide University. He is an internationally recognised ophthalmic clinician/scientist with special interests in glaucoma, retinal cell biology, neuroprotection, ophthalmic epidemiology and translational ophthalmic research.
Aim of the project
The retinal ganglion cells (RGCs) are the cells at the back of the eye that degenerate in glaucoma: we are investigating photobiomodulation (PBM) as a novel therapy to prevent loss of these cells.
Methods
We are assessing PBM in cell culture models and in animal models of glaucoma.
Key Results
To date, we have demonstrated a protective effect of PBM in our cell culture models and have shown that PBM influences the energy metabolism of the cell by acting on the cell’s powerhouse: the mitochondria.
Conclusions
PBM favourably influences mitochondrial function in retinal cells and protects cells from degeneration induced by metabolic stressors.
Clinical Implications
Based on previous ORIA-funded research, we have previously shown that PBM rescues light- sensitive retinal cells (photoreceptors). We demonstrated this effect in the lab and completed a Phase I first-in-human trial in patients with retinitis pigmentosa. The current research expands on this ORIA-funded work and has provided encouraging results indicating that PBM may be applicable to glaucoma. The next steps are to assess PBM in the animal models of glaucoma with a view to demonstrating safety and efficacy and proceeding to a Phase I clinical trial in glaucoma. If successful, the research has the potential to provide a novel treatment in addition to eye pressure lowering therapy in glaucoma.
Publications/Presentations
Presented at the Optic Nerve Meeting in Obergurgl December 2022.
Lay summary of outcomes
Low energy red light therapy shows promise as a new therapy for glaucoma.
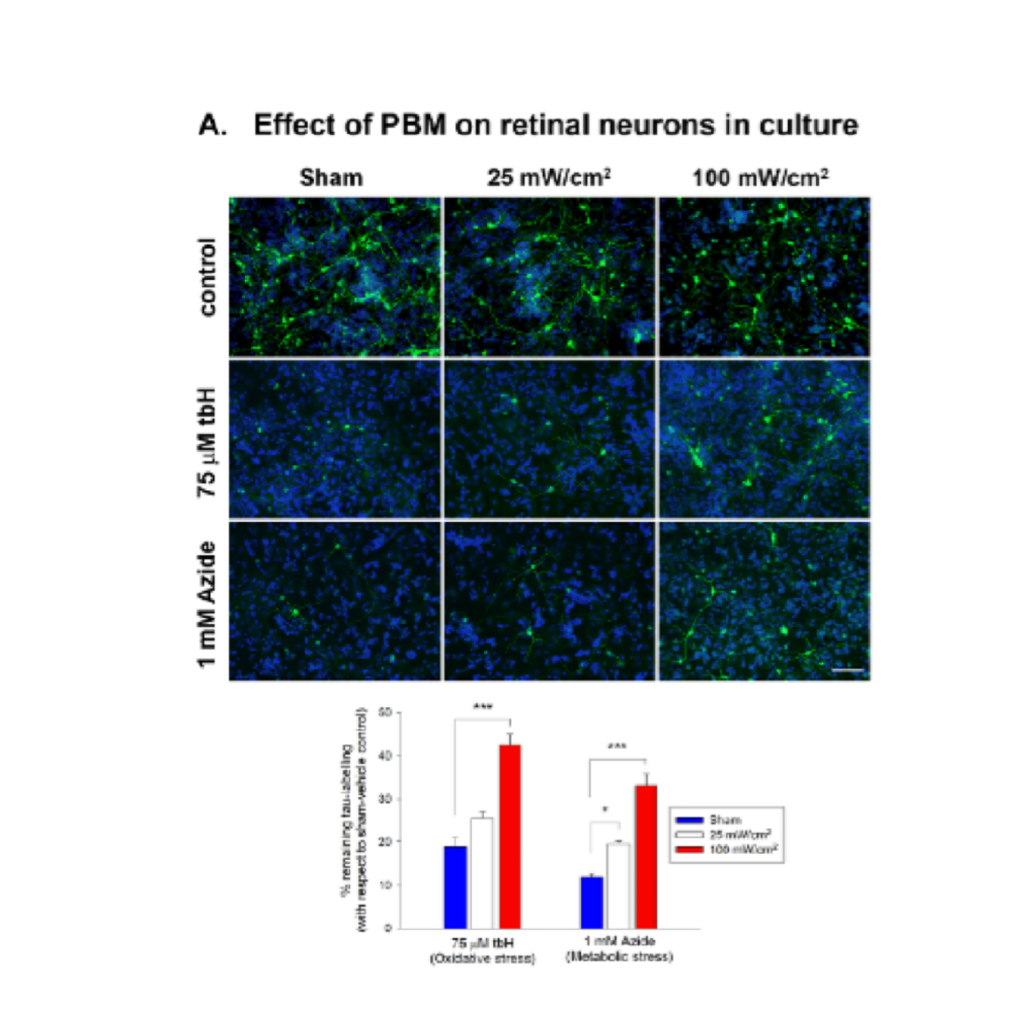
Fig. 1 Effect of PBM on retinal cultures. (A) Effect of PBM on survival of tau-immunolabelled retinal neurons subjected to oxidative stress (tbH) or metabolic stress (sodium azide). PBM laser application at both 25 and 100 mW/cm2 augmented survival of retinal neurons in both injury settings. Scale bar: 50 μm. Data are expressed as mean ± SEM. ***p<0.001, *p<0.05 by one-way ANOVA plus Tukey’s post hoc test, where n=6 determinations, from separate cultures.